
Sciton Laser Systems: The Clinical Standard in Aesthetic Medicine
FDA-cleared platforms engineered for measurable outcomes. Trusted by 8,000+ practitioners across 70+ countries for 29 years of evidence-based performance.

FDA-cleared platforms engineered for measurable outcomes. Trusted by 8,000+ practitioners across 70+ countries for 29 years of evidence-based performance.
"After evaluating five competing platforms in 2022, we selected the JOULE X with BBL HERO and HALO modules. Within 8 months, our skin resurfacing procedure volume increased 42% and patient-reported satisfaction scores rose from 7.8 to 9.3 on our 10-point scale. The dual-wavelength flexibility eliminated the need for a second standalone device, saving approximately $120,000 in capital expenditure."
"We integrated the BBL HERO into our vascular treatment program in March 2023. For rosacea patients specifically, our treatment time per session dropped from 35 minutes to under 12 minutes, and we observed 78% clearance rates after 3 sessions versus 55% with our previous IPL system. The CoolComfort feature reduced post-treatment erythema duration by roughly 40%, which our patients noticed immediately."
Each system is purpose-built with proprietary wavelength technology, rigorously validated through peer-reviewed clinical trials, and optimized for specific tissue targets and treatment protocols.

The most powerful broadband light platform available. Delivers 4x speed and 3x peak power for photorejuvenation, vascular treatments, and pigment correction with CoolComfort temperature management.
Request Specifications →
The first hybrid fractional laser combining non-ablative 1470nm and ablative 2940nm wavelengths. Independently tunable for precise depth control in skin resurfacing with minimal downtime protocols.
Request Specifications →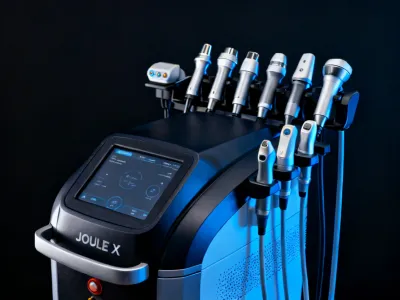
Modular platform accommodating multiple wavelengths and delivery systems in a single chassis. Expandable architecture supports Er:YAG ablative, Nd:YAG vascular, and BBL HERO modules for comprehensive practice coverage.
Request Specifications →Positioning accuracy to ±0.01mm with proprietary dual-wavelength Er:YAG and Nd:YAG platforms. Tissue-selective treatment depths from 10 to 350 microns with repeatable uniformity verified through pre-clinical testing.
147-point calibration and quality verification sequence before each shipment. ISO 13485 manufacturing standards with FDA-registered component sourcing throughout the supply chain.
R&D team of biomedical engineers, physicists, and clinical specialists with combined 400+ years of laser-tissue interaction expertise. Active research partnerships with university medical centers.
Validated treatment protocols across dermatology, plastic surgery, and aesthetic medicine specialties.
Ablative and non-ablative fractional treatments
BBL-based pigment and vascular correction
Nd:YAG targeting for vascular lesions
Non-invasive tissue remodeling protocols
Our specialists will evaluate your practice requirements and recommend the optimal platform configuration for your clinical workflow and patient demographics.